Jan 26, 2026
Medical Education on Generics: Do Doctors Learn Equivalence?
Tag: Hatch-Waxman Act

FDA Authorization of Generics: Legal Basis and Approval Process
Read More
The FDA approves generic drugs through a strict legal process based on the Hatch-Waxman Act. Generics must match brand drugs in active ingredients, strength, and bioequivalence - not just cost. Over 90% of U.S. prescriptions are for generics, saving billions annually.
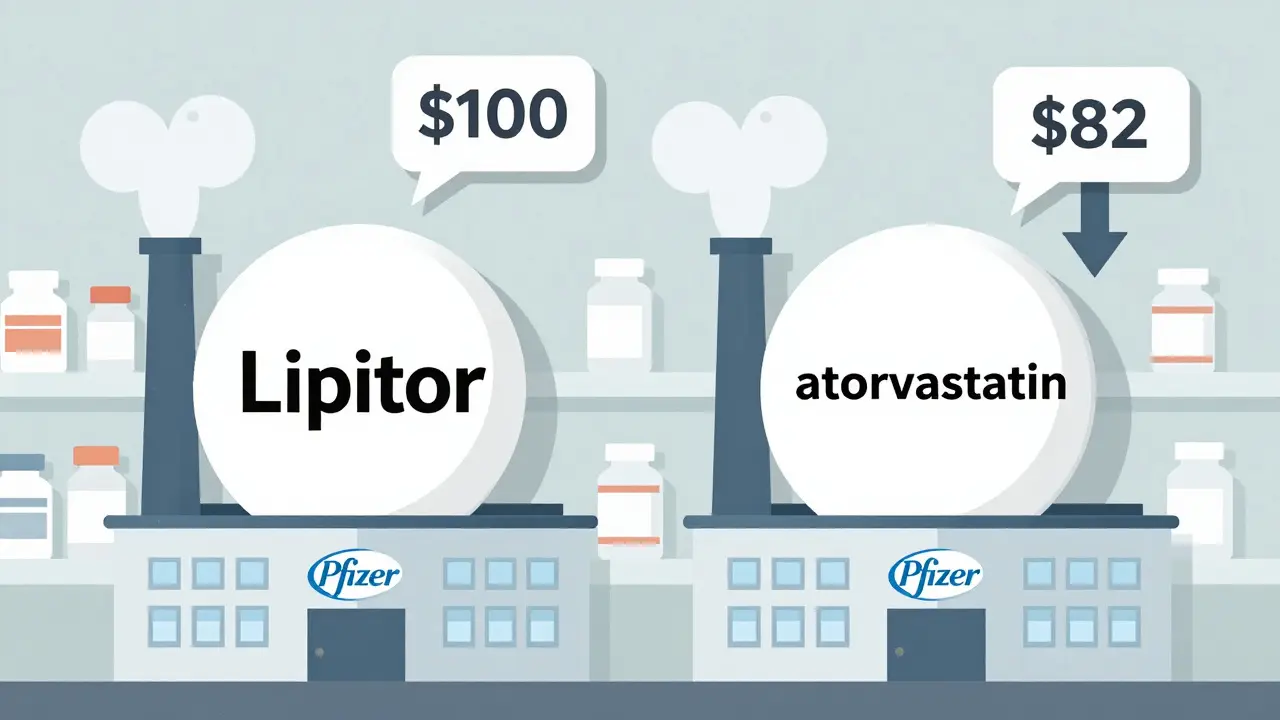
Cost Comparison: Authorized Generics vs First-to-File Generics
Read More
Authorized generics and first-to-file generics both lower drug costs, but when they compete, prices drop even more. Learn how these two types of generics affect your prescription bills and why competition matters.





